
Back آزاثيوبرين Arabic آزاتیوپرین AZB Azatioprina Catalan Asathioprin Welsh Azathioprin German Αζαθειοπρίνη Greek Azatioprina Spanish Azatioprina Basque آزاتیوپرین Persian Atsatiopriini Finnish
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /ˌæzəˈθaɪəˌpriːn/[1] |
| Trade names | Azasan, Imuran, Jayempi, others |
| Other names | AZA |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682167 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth, intravenous |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 60±31% |
| Protein binding | 20–30% |
| Metabolism | Activated non-enzymatically, deactivated mainly by xanthine oxidase |
| Elimination half-life | 26–80 minutes (azathioprine) 3–5 hours (drug plus metabolites) |
| Excretion | Kidney, 98% as metabolites |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.006.525 |
| Chemical and physical data | |
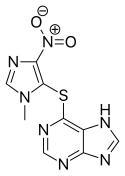
| Formula | C9H7N7O2S |
| Molar mass | 277.26 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 238 to 245 °C (460 to 473 °F) |
| |
| |
| (verify) | |
Azathioprine, sold under the brand name Imuran, among others, is an immunosuppressive medication.[5] It is used for the treatment of rheumatoid arthritis, granulomatosis with polyangiitis, Crohn's disease, ulcerative colitis, and systemic lupus erythematosus, and in kidney transplants to prevent rejection. It is listed by the International Agency for Research on Cancer as a group 1 human carcinogen.[5][6][7][8] It is taken by mouth or injected into a vein.[5]
Common side effects include bone-marrow suppression and vomiting.[5] Bone-marrow suppression is especially common in people with a genetic deficiency of the enzyme thiopurine S-methyltransferase.[5] Other serious risk factors include an increased risk of certain cancers.[5] Use during pregnancy may result in harm to the baby.[5] Azathioprine belongs to the purine analogues subclass of antimetabolites family of medications.[5][9] It works via 6-thioguanine to disrupt the making of RNA and DNA by cells.[5][9]
Azathioprine was first made in 1957.[9] It is on the World Health Organization's List of Essential Medicines.[10] In 2018, it was the 358th-most commonly prescribed medication in the United States, with more than 800,000 prescriptions.[11]
- ^ "Azathioprine". Merriam-Webster.com Dictionary. Merriam-Webster.
- ^ "FDA-sourced list of all drugs with black box warnings (Use Download Full Results and View Query links.)". nctr-crs.fda.gov. FDA. Retrieved 22 Oct 2023.
- ^ "Jayempi EPAR". European Medicines Agency. 20 April 2021. Retrieved 4 March 2023.
- ^ "Jayempi Product information". Union Register of medicinal products. Retrieved 3 March 2023.
- ^ a b c d e f g h i "Azathioprine". The American Society of Health-System Pharmacists. Archived from the original on 20 August 2016. Retrieved 8 December 2016.
- ^ Axelrad JE, Lichtiger S, Yajnik V (May 2016). "Inflammatory bowel disease and cancer: The role of inflammation, immunosuppression, and cancer treatment". World Journal of Gastroenterology (Review). 22 (20): 4794–4801. doi:10.3748/wjg.v22.i20.4794. PMC 4873872. PMID 27239106.
- ^ Singer O, McCune WJ (May 2017). "Update on maintenance therapy for granulomatosis with polyangiitis and microscopic polyangiitis". Current Opinion in Rheumatology. 29 (3): 248–253. doi:10.1097/BOR.0000000000000382. PMID 28306595. S2CID 35805200.
- ^ Jordan N, D'Cruz D (2016). "Current and emerging treatment options in the management of lupus". ImmunoTargets and Therapy. 5: 9–20. doi:10.2147/ITT.S40675. PMC 4970629. PMID 27529058.
- ^ a b c Sami N (2016). Autoimmune Bullous Diseases: Approach and Management. Springer. p. 83. ISBN 9783319267289. Archived from the original on 2016-12-21.
- ^ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ "Azathioprine - Drug Usage Statistics". ClinCalc. Retrieved 7 October 2022.