
Back Kernfusie Afrikaans Fusión nucleyar AN اندماج نووي Arabic تدماج نووي ARY Fusión nuclear AST Termonüvə reaksiyası Azerbaijani فوزیون AZB Тэрмаядзерная рэакцыя Byelorussian Термоядрен синтез Bulgarian কেন্দ্রীণ সংযোজন Bengali/Bangla
| Nuclear physics |
|---|
 |
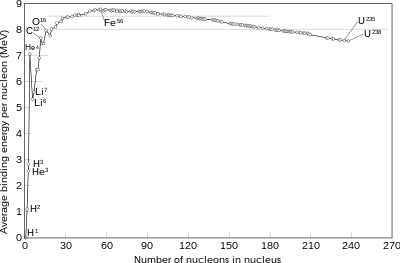
Nuclear fusion is a reaction in which two or more atomic nuclei (for example, nuclei of hydrogen isotopes deuterium and tritium), combine to form one or more atomic nuclei and neutrons. The difference in mass between the reactants and products is manifested as either the release or absorption of energy. This difference in mass arises as a result of the difference in nuclear binding energy between the atomic nuclei before and after the fusion reaction. Nuclear fusion is the process that powers active or main-sequence stars and other high-magnitude stars, where large amounts of energy are released.
A nuclear fusion process that produces atomic nuclei lighter than iron-56 or nickel-62 will generally release energy. These elements have a relatively small mass and a relatively large binding energy per nucleon. Fusion of nuclei lighter than these releases energy (an exothermic process), while the fusion of heavier nuclei results in energy retained by the product nucleons, and the resulting reaction is endothermic. The opposite is true for the reverse process, called nuclear fission. Nuclear fusion uses lighter elements, such as hydrogen and helium, which are in general more fusible; while the heavier elements, such as uranium, thorium and plutonium, are more fissionable. The extreme astrophysical event of a supernova can produce enough energy to fuse nuclei into elements heavier than iron.
- ^ "Ask Us: Sun". Cosmicopia. NASA. 2012. Archived from the original on 3 September 2018. Retrieved 13 July 2017.

