
Back نيستاتين Arabic Nistatin Azerbaijani نیستاتین AZB Nistatina Catalan Nystatin Welsh Nystatin German Νυστατίνη Greek Nistatina Spanish نیستاتین Persian Nystatiini Finnish
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Mycostatin, others[1] |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682758 |
| License data | |
| Routes of administration | Topical, vaginal, by mouth |
| Drug class | Polyene antifungal medication[1] |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 0% on oral ingestion |
| Metabolism | None (not extensively absorbed) |
| Elimination half-life | Dependent upon GI transit time |
| Excretion | Fecal (100%) |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| NIAID ChemDB | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.014.317 |
| Chemical and physical data | |
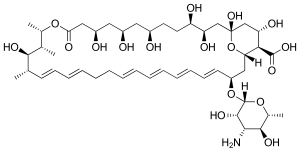
| Formula | C47H75NO17 |
| Molar mass | 926.107 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 44–46 °C (111–115 °F) |
| |
| |
| | |
Nystatin, sold under the brand name Mycostatin among others, is an antifungal medication.[1] It is used to treat Candida infections of the skin including diaper rash, thrush, esophageal candidiasis, and vaginal yeast infections.[1] It may also be used to prevent candidiasis in those who are at high risk.[1] Nystatin may be used by mouth, in the vagina, or applied to the skin.[1]
Common side effects when applied to the skin include burning, itching, and a rash.[1] Common side effects when taken by mouth include vomiting and diarrhea.[1] During pregnancy use in the vagina is safe while other formulations have not been studied in this group.[1] It works by disrupting the cell membrane of the fungal cells.[1]
Nystatin was discovered in 1950 by Rachel Fuller Brown and Elizabeth Lee Hazen.[2] It was the first polyene macrolide antifungal.[3] It is on the World Health Organization's List of Essential Medicines.[4] It is available as a generic medication.[1] It is made from the bacterium Streptomyces noursei.[2] In 2022, it was the 236th most commonly prescribed medication in the United States, with more than 1 million prescriptions.[5][6]
- ^ a b c d e f g h i j k "Nystatin". American Society of Health-System Pharmacists. Archived from the original on 3 February 2016. Retrieved 27 January 2016.
- ^ a b Espinel-Ingroff AV (2013). Medical Mycology in the United States a Historical Analysis (1894-1996). Dordrecht: Springer Netherlands. p. 62. ISBN 9789401703116. Archived from the original on 2 February 2016.
- ^ Gupte M, Kulkarni P, Ganguli BN (January 2002). "Antifungal antibiotics". Applied Microbiology and Biotechnology. 58 (1): 46–57. doi:10.1007/s002530100822. PMID 11831475. S2CID 8015426.
- ^ World Health Organization (2023). The selection and use of essential medicines 2023: web annex A: World Health Organization model list of essential medicines: 23rd list (2023). Geneva: World Health Organization. hdl:10665/371090. WHO/MHP/HPS/EML/2023.02.
- ^ "The Top 300 of 2022". ClinCalc. Archived from the original on 30 August 2024. Retrieved 30 August 2024.
- ^ "Nystatin Drug Usage Statistics, United States, 2013 - 2022". ClinCalc. Retrieved 30 August 2024.