
Back فيبوكسوستات Arabic Febuxostat German Φεμπουξοστάτη Greek Febuxostat Spanish فبوکسوستات Persian Febuxostat French Febuksostat ID Febuxostat Italian フェブキソスタット Japanese ଫେବୁକ୍ସୋସ୍ଟାଟ OR
 | |
| Clinical data | |
|---|---|
| Trade names | Uloric, Adenuric, others[1] |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a609020 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | ≥84% absorbed |
| Protein binding | 99.2% to albumin |
| Metabolism | via CYP1A1, 1A2, 2C8, 2C9, UGT1A1, 1A8, 1A9[6] |
| Elimination half-life | ~5–8 hours |
| Excretion | Urine (~49%, mostly as metabolites, 3% as unchanged drug); feces (~45%, mostly as metabolites, 12% as unchanged drug) |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.207.329 |
| Chemical and physical data | |
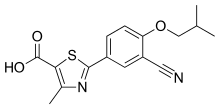
| Formula | C16H16N2O3S |
| Molar mass | 316.38 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Febuxostat, sold under the brand name Uloric among others, is a medication used long-term to treat gout due to high uric acid levels.[7] It is generally recommended only for people who cannot take allopurinol.[8][9] It is taken by mouth.[7]
Common side effects include liver problems, nausea, joint pain, and a rash.[7] Serious side effects include an increased risk of death as compared with allopurinol, Stevens–Johnson syndrome, and anaphylaxis.[9][8] Use is not recommended during pregnancy or breastfeeding.[9] It inhibits xanthine oxidase, thus reducing production of uric acid in the body.[7]
Febuxostat was approved for medical use in the European Union in 2008,[5] and in the United States in 2009.[7] A generic version was approved in 2019.[10][11]
- ^ Cite error: The named reference
drugnameswas invoked but never defined (see the help page). - ^ "Febuxostat (Uloric) Use During Pregnancy". Drugs.com. 22 February 2019. Retrieved 17 May 2020.
- ^ "FDA-sourced list of all drugs with black box warnings (Use Download Full Results and View Query links.)". nctr-crs.fda.gov. FDA. Retrieved 22 October 2023.
- ^ "Prescription medicines: registration of new chemical entities in Australia, 2014". Therapeutic Goods Administration (TGA). 21 June 2022. Retrieved 10 April 2023.
- ^ a b "Adenuric EPAR". European Medicines Agency (EMA). 21 April 2008. Retrieved 14 September 2024.
- ^ Cite error: The named reference
EMAwas invoked but never defined (see the help page). - ^ a b c d e "Febuxostat Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Retrieved 26 February 2019.
- ^ a b "Drug Safety and Availability - FDA adds Boxed Warning for increased risk of death with gout medicine Uloric (febuxostat)". FDA. 21 February 2019. Retrieved 26 February 2019.
- ^ a b c British national formulary : BNF 76 (76 ed.). Pharmaceutical Press. 2018. p. 1087. ISBN 9780857113382.
- ^ "Generic Uloric Availability". Drugs.com. Retrieved 1 August 2019.
- ^ "Febuxostat Generic Uloric". Retrieved 15 April 2020.